
UCB invests as much as $2.2 billion in Candid Therapeutics, reinforcing its commitment to T-cell engagers for treating autoimmune diseases.
Belgian pharmaceutical company UCB is acquiring a San Diego biotech firm, Candid Therapeutics, which is two years old, for an upfront payment of $2 billion. This marks UCB's second significant investment in T-cell engagers within a few months, based on the premise that cancer-targeting B-cell therapies can transform the treatment of autoimmune diseases.
Candid, which has no approved drugs yet, has conducted trials on its lead program involving around 100 patients in various early-stage studies. The deal, announced on May 3, includes $2 billion in immediate cash and up to $200 million in milestone payments contingent upon development and regulatory achievements.
The acquisition is expected to finalize by the end of the second quarter or early in the third, pending antitrust approvals. UCB has maintained its financial guidance for 2026, indicating that it aims to absorb this acquisition without altering its expectations.
This is UCB’s second recent pursuit of the same therapeutic approach, having previously licensed ATG-201, a bispecific from China's Antengene, for up to $1.1 billion. The Candid acquisition builds on this and introduces a different target related to B-cells.
Candid’s main asset, cizutamig, is a bispecific antibody designed to connect T-cells and plasma cells, targeting the latter to promote destruction. This mechanism was developed for the treatment of multiple myeloma, where eliminating rogue plasma cells is crucial. UCB believes that this mechanism can also be adapted to eliminate autoreactive B-cells in autoimmune diseases such as lupus and myasthenia gravis.
According to UCB, cizutamig has been clinically assessed in over 100 patients for both multiple myeloma and autoimmune disorders, and it is currently in Phase 1 studies for more than ten autoimmune conditions. UCB describes it as a potential leading agent in the BCMA T-cell engager category for autoimmune disease—a statement that conveys both ambition and typical corporate language.
The willingness of buyers to invest significant sums based on Phase 1 data stems from the encouraging early signals observed in autoimmune treatments. Some patients with severe conditions have experienced lasting remissions after a single cycle of B-cell-depleting therapy, even where conventional treatments have provided only partial relief. However, definitive results will require more robust late-stage data, larger patient groups, and extended follow-up.
Candid, established in 2024 with $370 million in initial funding from Two River Group and Vida Ventures, is led by Dr. Ken Song, who previously guided RayzeBio through its $4.1 billion acquisition by Bristol Myers Squibb. His track record in building and selling clinical-stage oncology and immunology companies contributes to the value UCB recognizes in Candid.
The disparity between Candid's launch funding and the $2 billion upfront payment from UCB illustrates investor confidence in the company's potential under Dr. Song's leadership. Earlier this year, Candid planned to go public through a reverse merger with Rallybio, but this strategy appears to have been overtaken by UCB's more appealing offer.
UCB's acquisition reflects a broader trend wherein major pharmaceutical firms are investing in T-cell engagers targeting autoimmune diseases. Over the past several months, several companies, including Gilead and Sanofi, have made significant investments in similar therapeutic platforms.
The urgency behind these deals can be attributed to two factors: the applicability of oncology science for autoimmune treatments, and the vast market potential for successful autoimmune therapies. If T-cell engagers demonstrate efficacy in these conditions, the financial rewards could be substantial, whereas unsuccessful ventures may face scrutiny for their high costs.
It's important to note what isn’t driving this acquisition. Despite the growing interest in AI-driven drug discovery, cizutamig was developed through traditional methods, not generative protein models. Current autoimmune deal flows predominantly consist of compounds developed through conventional chemistry from the previous decade, indicating that immediate value creation is occurring through established entities rather than AI.
For UCB, the acquisition serves a strategic purpose. With its established immunology division and history of expanding through targeted mergers, combining Candid’s BCMAxCD3 asset with Antengene’s CD19xCD3 candidate enhances its portfolio of B-cell-depleting therapies in a competitive market favoring diverse therapeutic approaches.
UCB's challenge lies in execution. While Phase 1 data is promising, the market is crowded with competitors, and any successful T-cell engager will likely face pricing pressures as it approaches market approval. Additionally, scaling up the manufacturing of bispecific antibodies poses its own challenges.
As the deal progresses, the market's response to T-cell engagers may evolve. UCB's proactive stance may prove beneficial; however, the true test will emerge through the Phase 2 results expected over the next 18 months. For now, a fledgling company backed by experienced leadership has attracted a $2.2 billion investment from a company that believes in its potential.
Otros artículos
 Plex is experiencing a significant increase in price for remote streaming.
Plex will conclude the promotional pricing for its Remote Watch Pass on June 1, increasing the monthly cost to $2.99 and the annual cost to $29.99.
Plex is experiencing a significant increase in price for remote streaming.
Plex will conclude the promotional pricing for its Remote Watch Pass on June 1, increasing the monthly cost to $2.99 and the annual cost to $29.99.
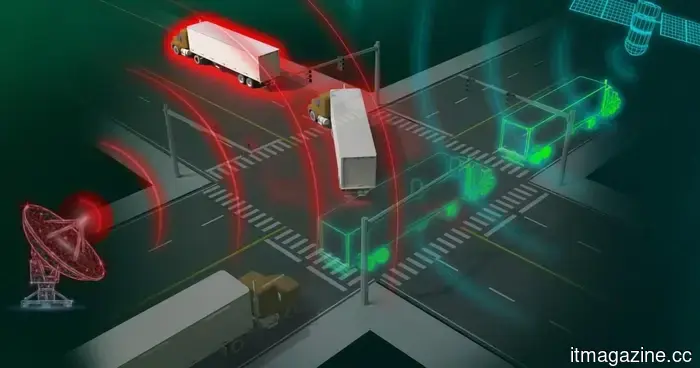 The renowned Oak Ridge laboratory has just created a portable device capable of detecting GPS spoofing in real time.
GPS spoofing enables criminals to steal trucks, hijack shipments, and redirect hazardous cargo, all while maintaining the appearance of normalcy. A newly developed detector from Oak Ridge National Lab is the first to identify this activity in real time.
The renowned Oak Ridge laboratory has just created a portable device capable of detecting GPS spoofing in real time.
GPS spoofing enables criminals to steal trucks, hijack shipments, and redirect hazardous cargo, all while maintaining the appearance of normalcy. A newly developed detector from Oak Ridge National Lab is the first to identify this activity in real time.
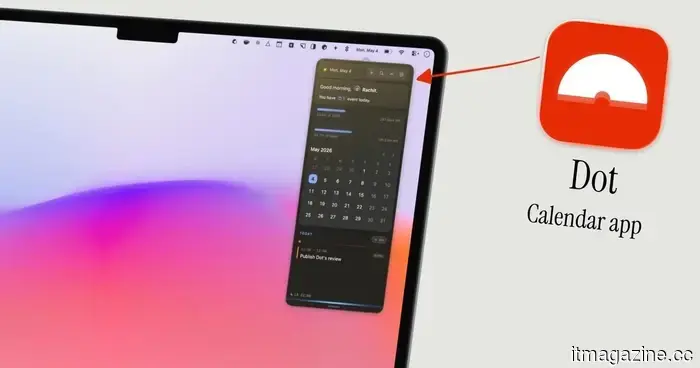 Dot is the Mac calendar application I wish I had discovered earlier in 2026.
I've always enjoyed using menu bar calendar applications. They allow me to quickly view upcoming events, add new ones effortlessly, and access my calendar from anywhere. Dot is the best one I've encountered.
Dot is the Mac calendar application I wish I had discovered earlier in 2026.
I've always enjoyed using menu bar calendar applications. They allow me to quickly view upcoming events, add new ones effortlessly, and access my calendar from anywhere. Dot is the best one I've encountered.
 UCB is investing up to $2.2 billion to acquire Candid Therapeutics, reinforcing its commitment to T-cell engagers in the area of autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, focusing on cizutamig and the growing trend of T-cell engagers in autoimmune diseases.
UCB is investing up to $2.2 billion to acquire Candid Therapeutics, reinforcing its commitment to T-cell engagers in the area of autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, focusing on cizutamig and the growing trend of T-cell engagers in autoimmune diseases.
 Microsoft Edge is eliminating sidebar applications as part of the ongoing effort to declutter Windows 11.
Microsoft is discontinuing the sidebar app list in Edge as part of its effort to simplify the browser, eliminating pinned web applications but retaining Copilot.
Microsoft Edge is eliminating sidebar applications as part of the ongoing effort to declutter Windows 11.
Microsoft is discontinuing the sidebar app list in Edge as part of its effort to simplify the browser, eliminating pinned web applications but retaining Copilot.
 Samsung's forthcoming Galaxy Z Flip 8 could potentially resolve a significant display issue.
Samsung's Galaxy Z Flip 8 appears to be a significant yet understated upgrade, featuring a long-awaited solution for the crease-free display alongside a series of minor hardware enhancements.
Samsung's forthcoming Galaxy Z Flip 8 could potentially resolve a significant display issue.
Samsung's Galaxy Z Flip 8 appears to be a significant yet understated upgrade, featuring a long-awaited solution for the crease-free display alongside a series of minor hardware enhancements.
UCB invests as much as $2.2 billion in Candid Therapeutics, reinforcing its commitment to T-cell engagers for treating autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, placing its confidence in cizutamig and the broader surge of T-cell engagers for autoimmune diseases.
