
UCB is investing up to $2.2 billion to acquire Candid Therapeutics, reinforcing its commitment to T-cell engagers in the area of autoimmune diseases.
The Belgian pharmaceutical company is acquiring a two-year-old biotech firm based in San Diego for $2 billion upfront, marking its second investment in T-cell engagers (TCE) in recent months. The underlying idea is that B-cell-targeting therapies designed for cancer could transform the treatment of autoimmune diseases.
Candid Therapeutics, which is just two years old, has not yet received approval for any drugs. Its primary program has been evaluated in about 100 patients in various early-stage trials. On Sunday, UCB, the pharmaceutical company listed in Brussels, announced its decision to purchase Candid for a total of up to $2.2 billion.
Such a valuation for a biotech firm at this stage warrants clarification, which is linked to the potential of T-cell engagers in treating autoimmune diseases by 2026. According to the agreement disclosed on May 3, UCB will make an upfront cash payment of $2 billion, plus up to $200 million in additional payments contingent on development and regulatory advancements.
The deal is anticipated to be finalized by the end of the second quarter or early in the third, pending antitrust approval. UCB has confirmed its financial projections for 2026, indicating that it plans to integrate this acquisition without altering its outlook.
This is the second instance in recent months where the Belgian company has invested in the same therapeutic mechanism. Previously, UCB licensed ATG-201, a bispecific antibody targeting CD19/CD3 from the China-based company Antengene, in a deal valued at up to $1.1 billion. The acquisition of Candid adds to this strategy, focusing on a different B-cell target.
What does Candid possess?
Candid’s primary asset is cizutamig, a bispecific antibody engineered to connect two different cells: it binds to a T-cell using CD3 and to a plasma cell through BCMA, the B-cell maturation antigen, instructing the T-cell to eliminate the plasma cell.
This mechanism was designed for the treatment of multiple myeloma, where the goal is to eliminate rogue plasma cells. The hypothesis for 2026 is that the same approach can be adapted to target autoreactive B-cells and plasma cells involved in autoimmune diseases like lupus and myasthenia gravis, as well as a variety of other less common conditions where the immune system attacks its own tissues.
UCB reports that cizutamig has been clinically assessed in over 100 patients across multiple myeloma and autoimmune indications and is currently undergoing Phase 1 trials for over ten autoimmune diseases. In its statement, UCB refers to it as a potential best-in-class BCMA T-cell engager for autoimmune diseases, which is a bold claim commonly seen in press releases of this nature.
The reason investors are willing to commit nine-figure sums based on Phase 1 data is due to the remarkably promising early results from this drug class in autoimmune contexts. Patients with severe conditions have demonstrated lasting remissions following just one round of B-cell-depleting treatment, even in cases where traditional small-molecule and biologic therapies have provided only limited success. However, definitive results remain pending.
Further late-stage data, larger patient groups, and extended follow-up will be necessary. Yet, the consistent positive direction of these preliminary results has led pharma companies to start valuing this approach as if it will be effective.
Candid was established in 2024 in San Diego, funded by Two River Group and Vida Ventures, with an initial financing of $370 million. Dr. Ken Song, who previously guided RayzeBio through its $4.1 billion acquisition by Bristol Myers Squibb in late 2023, serves as the company's chairman, CEO, and president. He has a track record of developing, scaling, and selling clinical-stage oncology and immunology biotechs.
This background contributes to UCB's willingness to invest. Buyers in this market segment increasingly consider management quality alongside the quality of the molecule itself, especially when a molecule's commercial potential hinges on well-structured trials across numerous minor indications.
The disparity in valuation between the initial $370 million in funding obtained in mid-2024 and the $2 billion upfront payment from UCB signifies the value investors attribute to what Dr. Song and his team have created.
This also marks a significant turnaround. In March, Candid had announced a reverse merger with Rallybio, a smaller publicly traded rare-disease company, aimed at facilitating Candid’s public listing. However, that transaction has seemingly been superseded by UCB’s more attractive offer.
UCB's acquisition aligns with a noticeable trend. Over the past nine months, every major pharma company with a presence in immunology has either acquired, licensed, or partnered in relation to T-cell engagers targeting autoimmune disease.
For instance, Gilead purchased Ouro Medicines for $2.18 billion earlier this year to acquire gamgertamig, another BCMAxCD3 engager. Sanofi licensed a tris
Other articles
 Microsoft Edge is eliminating sidebar applications as part of the ongoing effort to declutter Windows 11.
Microsoft is discontinuing the sidebar app list in Edge as part of its effort to simplify the browser, eliminating pinned web applications but retaining Copilot.
Microsoft Edge is eliminating sidebar applications as part of the ongoing effort to declutter Windows 11.
Microsoft is discontinuing the sidebar app list in Edge as part of its effort to simplify the browser, eliminating pinned web applications but retaining Copilot.
 Plex is experiencing a significant increase in prices for remote streaming services.
Plex will conclude the introductory pricing for its Remote Watch Pass on June 1, increasing monthly charges to $2.99 and annual charges to $29.99.
Plex is experiencing a significant increase in prices for remote streaming services.
Plex will conclude the introductory pricing for its Remote Watch Pass on June 1, increasing monthly charges to $2.99 and annual charges to $29.99.
 Plex is experiencing a significant increase in price for remote streaming.
Plex will conclude the promotional pricing for its Remote Watch Pass on June 1, increasing the monthly cost to $2.99 and the annual cost to $29.99.
Plex is experiencing a significant increase in price for remote streaming.
Plex will conclude the promotional pricing for its Remote Watch Pass on June 1, increasing the monthly cost to $2.99 and the annual cost to $29.99.
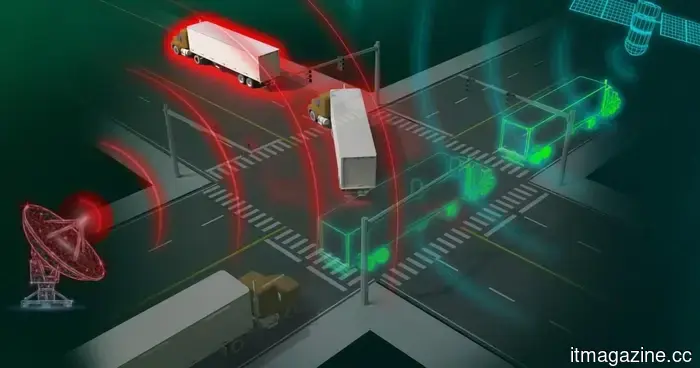 The renowned Oak Ridge laboratory has just created a portable device capable of detecting GPS spoofing in real time.
GPS spoofing enables criminals to steal trucks, hijack shipments, and redirect hazardous cargo, all while maintaining the appearance of normalcy. A newly developed detector from Oak Ridge National Lab is the first to identify this activity in real time.
The renowned Oak Ridge laboratory has just created a portable device capable of detecting GPS spoofing in real time.
GPS spoofing enables criminals to steal trucks, hijack shipments, and redirect hazardous cargo, all while maintaining the appearance of normalcy. A newly developed detector from Oak Ridge National Lab is the first to identify this activity in real time.
 China's rise in industry is propelled by commercialization rather than subsidies - and the West lacks a response to this.
China has invested $184 billion in AI venture capital, achieving 86% of its Made in China 2025 goals, with research and development expenditures exceeding those of the United States. The key factor is large-scale commercialization rather than government funding.
China's rise in industry is propelled by commercialization rather than subsidies - and the West lacks a response to this.
China has invested $184 billion in AI venture capital, achieving 86% of its Made in China 2025 goals, with research and development expenditures exceeding those of the United States. The key factor is large-scale commercialization rather than government funding.
 UCB invests as much as $2.2 billion in Candid Therapeutics, reinforcing its commitment to T-cell engagers for treating autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, placing its confidence in cizutamig and the broader surge of T-cell engagers for autoimmune diseases.
UCB invests as much as $2.2 billion in Candid Therapeutics, reinforcing its commitment to T-cell engagers for treating autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, placing its confidence in cizutamig and the broader surge of T-cell engagers for autoimmune diseases.
UCB is investing up to $2.2 billion to acquire Candid Therapeutics, reinforcing its commitment to T-cell engagers in the area of autoimmune diseases.
UCB is set to invest as much as $2.2 billion in Candid Therapeutics, focusing on cizutamig and the growing trend of T-cell engagers in autoimmune diseases.
