
ONWARD Medical secures €40.6 million to progress its spinal cord stimulation implant.
The neurotechnology firm from Eindhoven is utilizing the funds to sponsor its Empower BP pivotal trial, which evaluates the capability of its implantable ARC-IM® system in managing blood pressure fluctuations following spinal cord injury, in conjunction with the broadened commercial launch of its already approved ARC-EX® external therapy system. The financial runway is now extended to Q1 2028.
ONWARD Medical has secured €40.6 million in gross proceeds via an accelerated bookbuild private placement involving institutional investors, as announced by the Eindhoven-based neurotechnology company on April 16, 2026.
The deal was primarily backed by a €25 million investment from EQT Life Sciences, with additional participation from long-only and sector-specific investors. A total of 13,520,254 new ordinary shares were issued at €3.00 each.
The company’s shares are traded on Euronext Brussels, Amsterdam, and Paris under the ticker ONWD, with US ADRs available on OTCQX as ONWRY.
ONWARD is distributing the proceeds across four areas: 40% for the development of the investigational ARC-IM® implantable system, which includes clinical trials and regulatory activities; 30% for the expansion of sales and commercial operations for the ARC-EX® system in the US, Europe, and selected other markets; 20% for quality and administrative functions; and 10% for working capital, general corporate purposes, and servicing existing debt.
This additional capital, combined with current cash reserves, extends the company’s operations into Q1 2028, assuming there is no utilization of its debt facility.
The two products central to this fundraising represent consecutive phases of the company’s clinical progression. The ARC-EX® System, which is an external and non-invasive spinal cord stimulation therapy, has already received commercial clearance in both the United States and Europe.
It provides electrical stimulation to the spinal cord via surface electrodes, activating neural circuits below the injury to enhance voluntary motor function. The ARC-IM® System employs the same therapeutic principle but implements it deeper: it is an implantable device that offers stimulation directly, which ONWARD anticipates will yield more accurate and efficient treatment for various conditions, including blood pressure instability and bladder function, and when integrated with a brain-computer interface and AI, the restoration of movement through thought.
The Empower BP pivotal trial, currently the main focus of the development funding, is examining the ARC-IM®’s potential to manage orthostatic hypotension and autonomic dysreflexia, severe blood pressure issues that rank among the most debilitating secondary complications of spinal cord injury.
The trial received FDA Investigational Device Exemption approval in August 2025. It is randomized, double-blinded, and sham-controlled, representing the gold standard for pivotal trials of medical devices, and is expected to engage around 20 leading neurorehabilitation and neurosurgical research centers across the US, Canada, France, Germany, Spain, and the UK.
The first participant was enrolled and implanted in Q1 2026 at Craig Hospital in Denver, Colorado, with 10 sites now actively recruiting.
ONWARD holds 10 Breakthrough Device Designations from the US FDA, a status awarded to devices that offer more effective treatment for serious conditions with no satisfactory alternatives available. This designation accelerates the FDA review process and facilitates more frequent communication with the agency throughout development.
CEO Dave Marver articulated that this raise affirms both the commercial progress of ARC-EX® and the clinical promise of ARC-IM®: "The robust backing from EQT and other reputable investors highlights the swift adoption of the ARC-EX® System and confirms our successful shift to a commercial-stage organization."
Stifel Europe Securities SAS served as Sole Global Coordinator, with Bank Degroof Petercam SA/NV acting as joint bookrunner along with Stifel.
The new shares are anticipated to be listed on April 20, 2026. The company, EQT, and certain board members have committed to a 90-day lockup on the shares.

Other articles
 ONWARD Medical secures €40.6 million to further develop its spinal cord stimulation implant.
ONWARD Medical secures €40.6 million in a private placement led by EQT Life Sciences to finance its Empower BP pivotal trial and to enhance sales of ARC-EX.
ONWARD Medical secures €40.6 million to further develop its spinal cord stimulation implant.
ONWARD Medical secures €40.6 million in a private placement led by EQT Life Sciences to finance its Empower BP pivotal trial and to enhance sales of ARC-EX.
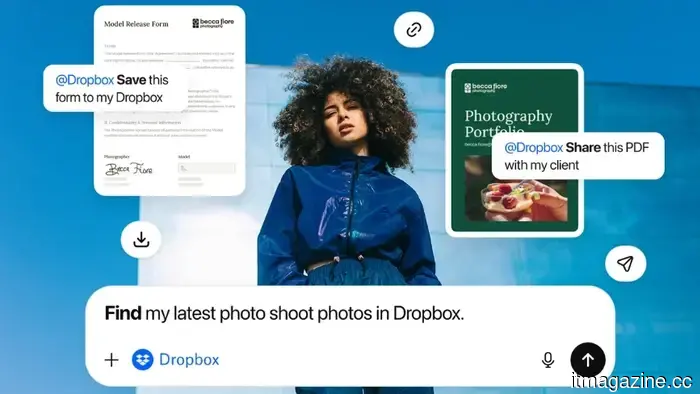 Dropbox is transforming ChatGPT into its central productivity platform by introducing three new applications.
Dropbox has introduced three ChatGPT applications that focus on file storage, enterprise search (Dash), and AI-driven calendar management (Reclaim).
Dropbox is transforming ChatGPT into its central productivity platform by introducing three new applications.
Dropbox has introduced three ChatGPT applications that focus on file storage, enterprise search (Dash), and AI-driven calendar management (Reclaim).
 Casely is recalling almost 500,000 power banks due to a fire risk. Here’s how to determine if your device is included in the recall.
The Casely Power Pods 5000mAh MagSafe wireless power bank is undergoing another recall. Here’s how to verify your unit and obtain a complimentary replacement.
Casely is recalling almost 500,000 power banks due to a fire risk. Here’s how to determine if your device is included in the recall.
The Casely Power Pods 5000mAh MagSafe wireless power bank is undergoing another recall. Here’s how to verify your unit and obtain a complimentary replacement.
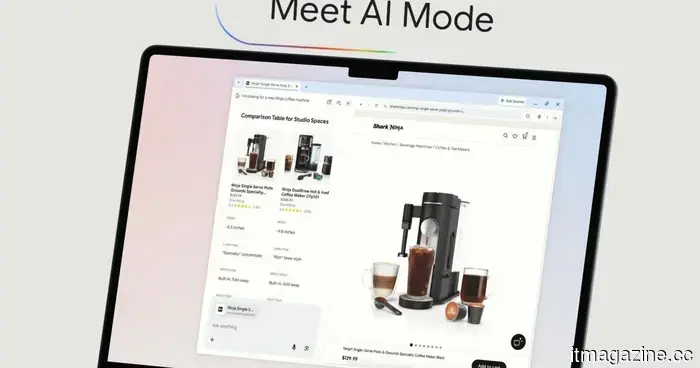 Amazon believes you are fond of AI, so it has introduced a dedicated storefront for gadgets powered by artificial intelligence.
In a world dominated by AI, determining what is effective becomes more complicated. This store simplifies the process.
Amazon believes you are fond of AI, so it has introduced a dedicated storefront for gadgets powered by artificial intelligence.
In a world dominated by AI, determining what is effective becomes more complicated. This store simplifies the process.
 The EU has granted its €180 million sovereign cloud contract.
The European Commission has granted a €180M sovereign cloud contract to Post Telecom, StackIT, Scaleway, and a consortium of Proximus, Thales, and Google Cloud, covering a period of six years.
The EU has granted its €180 million sovereign cloud contract.
The European Commission has granted a €180M sovereign cloud contract to Post Telecom, StackIT, Scaleway, and a consortium of Proximus, Thales, and Google Cloud, covering a period of six years.
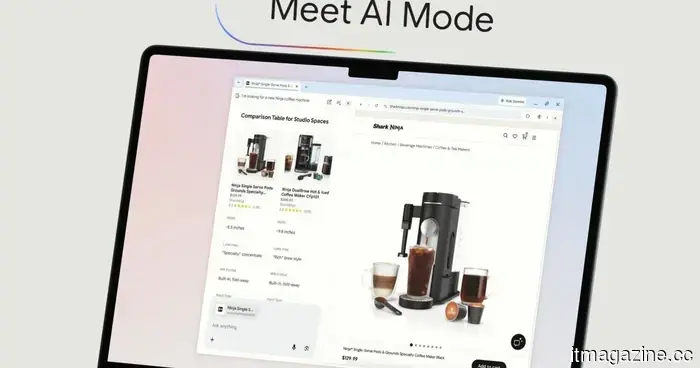 Amazon believes you have a fondness for AI, which is why it has introduced a dedicated storefront for AI-driven devices.
In a world dominated by AI, determining what is effective can be challenging. This store simplifies the process.
Amazon believes you have a fondness for AI, which is why it has introduced a dedicated storefront for AI-driven devices.
In a world dominated by AI, determining what is effective can be challenging. This store simplifies the process.
ONWARD Medical secures €40.6 million to progress its spinal cord stimulation implant.
ONWARD Medical secures €40.6M through a private placement led by EQT Life Sciences to finance its Empower BP pivotal trial and enhance ARC-EX commercial sales.
