Munich-based mbiomics secures €30 million to advance its live bacterial product.
The Munich-based techbio firm is working on a live bacterial product aimed at improving the response to immune checkpoint inhibitors in advanced melanoma, with a Phase 1B study scheduled for 2027. Mbiomics GmbH, specializing in microbiome-based therapeutics, has completed the final closing of its Series A funding round, achieving a total of €30 million raised. The latest €12 million funding was contributed by existing investors MIG Fonds and Bayern Kapital, marking the end of a round that commenced in March 2023.
The funds will support two immediate objectives: enhancing the IND-enabling pharmacological data package for the lead candidate MBX-116 and expediting the GMP-grade manufacturing development necessary for producing it at the clinical scale for human trials. The aim is to start a Phase 1B study in second-line advanced melanoma in 2027.
“While the clinical potential of the gut microbiome is well recognized, converting microbiome-based therapeutics into a scalable product has posed a significant engineering challenge,” stated Dr. Johannes B. Woehrstein, CEO and co-founder of mbiomics. “At mbiomics, we are addressing this challenge by developing a complete technology stack for the design, analysis, screening, and manufacturing of complex microbial consortia.”
Founded in Munich in 2020 by Woehrstein, Dr. Markus Rinecker, and Dr. Laura Figulla, mbiomics operates at the convergence of microbiology, AI-driven drug design, and precision medicine. Its main product category is Live Biotherapeutic Products (LBPs), which are oral therapeutics made from specific combinations of live bacterial strains, delivered in a pharmaceutical-grade format.
This approach is distinct from previous microbiome interventions: while fecal microbiota transplants (FMTs) have shown clinical efficacy in various conditions, they are inherently variable, non-standardized, and challenging to manufacture at scale. Mbiomics’ platform aims to eliminate the empirical variability of FMTs with a systematically designed, reproducible product.
The platform integrates AI and machine-learning-based consortia design, proprietary high-resolution analytical technology, and extensive co-cultivation and screening capabilities. The AI component is utilized not only for identifying candidate bacterial strains but also for designing the specific combinations of consortia likely to produce a targeted therapeutic effect in specific patient populations.
This method of rational consortia design, in contrast to single-strain probiotics or empirical transplants, positions mbiomics as a significant advancement from prior approaches. The company’s main clinical objective, MBX-116, is set to be used as a co-therapy with immune checkpoint inhibitors for second-line advanced melanoma, supported by a growing body of clinical evidence.
The gut microbiome influences the immune system in various ways: microbial metabolites, such as short-chain fatty acids and tryptophan-derived compounds, regulate immune cell activation, dendritic cell function, and the formation of regulatory T cells, collectively determining whether the immune system creates a sufficiently strong anti-tumor response.
Clinical evidence has shown a link between microbiome composition and responses to checkpoint inhibitors in multiple studies. A significant trial by Routy et al. revealed that FMT from donors who responded to immune checkpoint inhibitors substantially improved results in patients with refractory melanoma, with objective response rates hitting 65% in some groups. Conversely, patients who received broad-spectrum antibiotics within 30 days of starting checkpoint inhibitor treatment, which alters the gut microbiome, consistently exhibited poorer outcomes across various tumor types. Specific bacterial taxa, such as Akkermansia muciniphila and Faecalibacterium prausnitzii, have been consistently identified as more abundant in ICI responders and are mechanistically associated with enhanced effector T cell activity.
The total funding of €30 million is relatively modest compared to late-preclinical biotech rounds, but mbiomics is currently in the IND stage, completing the necessary pharmacological data package for filing an Investigational New Drug application with regulators. The 2027 target for the Phase 1B trial provides the company with approximately 18 months to achieve IND submission and begin the trial, a feasible timeline for a company at this stage if GMP manufacturing progresses as planned.
In addition to oncology, mbiomics has outlined a broader pipeline of microbiome-based therapeutics targeting conditions such as autoimmune and neurodegenerative diseases. These areas have garnered increasing research interest due to the connections between the gut-brain axis and gut-immune axis, yet no therapeutic-grade LBP has progressed to late-stage clinical development. Whether the company will pursue these indications independently or in partnership will likely depend on the outcomes of the melanoma trial.
Other articles
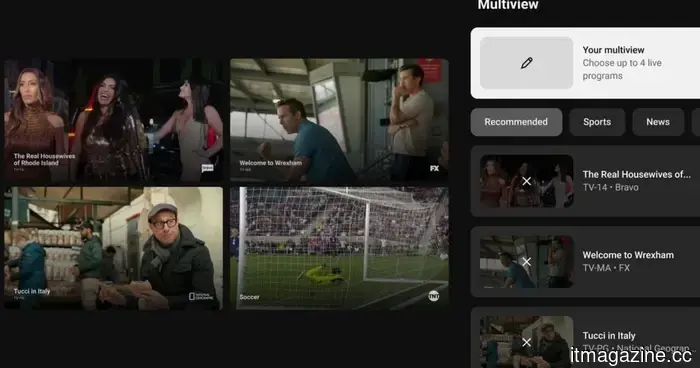 YouTube TV's custom multiview feature allows you to combine NFL Sunday Ticket with any other channel.
YouTube TV's personalized Multiview Builder is now operational, allowing subscribers to merge up to four live streams from any channel included in their subscription, such as NFL Sunday Ticket, across various categories.
YouTube TV's custom multiview feature allows you to combine NFL Sunday Ticket with any other channel.
YouTube TV's personalized Multiview Builder is now operational, allowing subscribers to merge up to four live streams from any channel included in their subscription, such as NFL Sunday Ticket, across various categories.
 The top headphone offers of 2026: substantial discounts on earbuds, studio headphones, and high-fidelity equipment.
Whether you're looking for true wireless earbuds for your daily commute or a pair of high-end headphones meant to last for years, this is an exceptionally good time to make a purchase. We're currently tracking discounts reaching up to 50% on some of the top-rated headphones available, including brands like Beats, Apple, Sennheiser, and HIFIMAN. [...]
The top headphone offers of 2026: substantial discounts on earbuds, studio headphones, and high-fidelity equipment.
Whether you're looking for true wireless earbuds for your daily commute or a pair of high-end headphones meant to last for years, this is an exceptionally good time to make a purchase. We're currently tracking discounts reaching up to 50% on some of the top-rated headphones available, including brands like Beats, Apple, Sennheiser, and HIFIMAN. [...]
 Netflix has confirmed the renewal of Stranger Things: Tales From '85 for a second season, even though it has received the lowest ratings in the franchise's history.
Despite receiving mixed reviews, Netflix has renewed Stranger Things: Tales From '85 for a second season. The animated spinoff currently has the lowest ratings for the franchise on both Rotten Tomatoes and IMDB.
Netflix has confirmed the renewal of Stranger Things: Tales From '85 for a second season, even though it has received the lowest ratings in the franchise's history.
Despite receiving mixed reviews, Netflix has renewed Stranger Things: Tales From '85 for a second season. The animated spinoff currently has the lowest ratings for the franchise on both Rotten Tomatoes and IMDB.
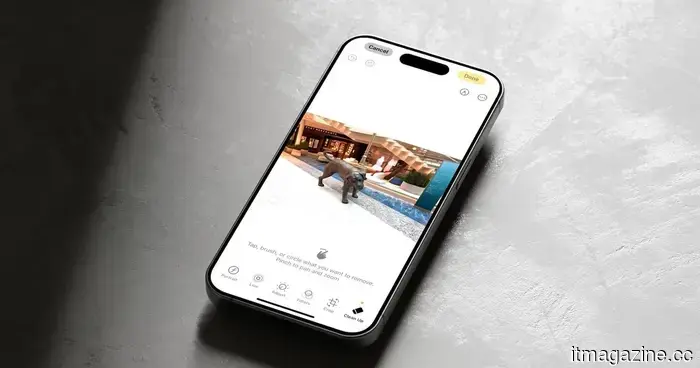 Apple is finally developing the AI photo editor that Google and Samsung have offered for years.
Enhance offers one-tap enhancements for quality, while Reframe modifies spatial perspectives in photos. However, both Extend and Reframe have encountered reliability problems during internal testing.
Apple is finally developing the AI photo editor that Google and Samsung have offered for years.
Enhance offers one-tap enhancements for quality, while Reframe modifies spatial perspectives in photos. However, both Extend and Reframe have encountered reliability problems during internal testing.
 Dex secures $5.3 million to develop an AI talent agency for engineers.
Dex, an AI-based talent agency for technical recruitment, has secured $5.3 million in funding, spearheaded by Notion Capital, and currently has an annual recurring revenue (ARR) of $1.8 million.
Munich-based mbiomics secures €30 million to advance the development of a live bacterial product.
Mbiomics has successfully completed a €30 million Series A funding round to further develop MBX-116, a live bacterial product aimed at enhancing the response to immune checkpoint inhibitors in patients with advanced melanoma.
Dex secures $5.3 million to develop an AI talent agency for engineers.
Dex, an AI-based talent agency for technical recruitment, has secured $5.3 million in funding, spearheaded by Notion Capital, and currently has an annual recurring revenue (ARR) of $1.8 million.
Munich-based mbiomics secures €30 million to advance the development of a live bacterial product.
Mbiomics has successfully completed a €30 million Series A funding round to further develop MBX-116, a live bacterial product aimed at enhancing the response to immune checkpoint inhibitors in patients with advanced melanoma.
Munich-based mbiomics secures €30 million to advance its live bacterial product.
Mbiomics has successfully finalized a €30 million Series A funding round to further develop MBX-116, a live bacterial product aimed at enhancing the response to immune checkpoint inhibitors in patients with advanced melanoma.
