
AliveCor’s Kardia 12L has received its CE marking.
The Kardia 12L has received the CE Mark, allowing it to launch in Europe. This device utilizes five electrodes and a single cable, replacing the cumbersome 10-lead ECG carts. Its AI system is capable of detecting 35 cardiac conditions, including acute myocardial infarction, and will be available initially in France, Germany, Italy, Spain, and the UK.
AliveCor, the US-based medical technology firm focused on AI-driven cardiac diagnostics, has secured the CE Mark for the Kardia 12L ECG system. Referred to as the world’s first AI-powered, portable 12-lead ECG with a distinct single-cable design, it will first be offered to healthcare professionals in the mentioned European countries, with plans to extend to more nations afterwards. The CE Mark enables distribution throughout the entire European Economic Area.
The Kardia 12L substitutes the standard 10-electrode ECG cart with a system using only five electrodes and a single cable, weighing just 0.13 kg. Its AI engine, KAI 12L, can identify 35 cardiac conditions, featuring 14 arrhythmias and 21 morphologies, including acute myocardial infarction and the most prevalent types of cardiac ischaemia.
This device is compact, battery-operated, and intended for contexts such as primary care, urgent care, pharmacies, rural clinics, and home visits by healthcare professionals, where traditional ECG carts are often too bulky and complicated to set up. The five-electrode configuration also allows patients to remain partially clothed during readings. A study published in Heart Rhythm O2 found that ECG acquisition time was reduced by nearly 30% compared to standard 12-lead systems.
Since its FDA clearance and subsequent commercial launch in the US in June 2024, the Kardia 12L has gained traction across the country and has later expanded to India, Australia, New Zealand, Vietnam, and Canada. The system has been utilized on tens of thousands of patients worldwide, identifying over 4,000 cases of myocardial infarction and ischaemia—figures AliveCor uses to highlight its clinical efficacy at scale.
The KAI 12L AI was trained and validated using more than 1.75 million ECGs from prominent US medical facilities. In January 2026, the FDA approved an updated version of KAI 12L that can detect 39 cardiac conditions, while the CE-marked version launches with 35, with the specific conditions varying by region as regulatory approvals are secured.
The timing of the European launch aligns with the EU Cardiovascular Health Plan, which emphasizes the bloc's commitment to early detection and better access to care for cardiovascular diseases, the leading cause of death in Europe. Simona Esposito, AliveCor’s Senior Vice President of Sales for Global Markets, described receiving the CE Mark as "a defining moment" for the company's international strategy, emphasizing that the device was designed for environments where traditional ECG carts would not be feasible.
AliveCor is a privately held firm based in Mountain View, California, and has recorded more than 350 million ECGs through its Kardia product lineup.
Other articles
 Sony's latest gaming monitor features a 720Hz refresh rate and is equipped with an OLED panel.
Developed in collaboration with Fnatic and utilizing LG's state-of-the-art tandem OLED technology, Sony's latest INZONE M10S II sets a new standard for the appearance, experience, and performance of competitive gaming monitors.
Sony's latest gaming monitor features a 720Hz refresh rate and is equipped with an OLED panel.
Developed in collaboration with Fnatic and utilizing LG's state-of-the-art tandem OLED technology, Sony's latest INZONE M10S II sets a new standard for the appearance, experience, and performance of competitive gaming monitors.
 OnePlus reveals an impressive gaming-focused phone that you probably won't be able to obtain.
You’re not prepared for this gaming powerhouse.
OnePlus reveals an impressive gaming-focused phone that you probably won't be able to obtain.
You’re not prepared for this gaming powerhouse.
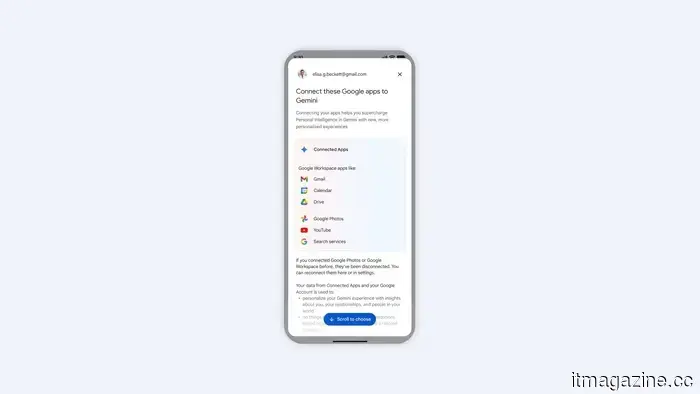 Google is broadening its Personal Intelligence feature to include Gemini users worldwide, marking a significant change.
Google is broadening the reach of Personal Intelligence for Gemini users worldwide, linking Gmail, Photos, YouTube, Maps, and other services to provide more intelligent, tailored responses without requiring additional prompts from you.
Google is broadening its Personal Intelligence feature to include Gemini users worldwide, marking a significant change.
Google is broadening the reach of Personal Intelligence for Gemini users worldwide, linking Gmail, Photos, YouTube, Maps, and other services to provide more intelligent, tailored responses without requiring additional prompts from you.
 OnePlus showcases an impressive gaming-focused smartphone that you probably won't be able to obtain.
You're not prepared for this gaming powerhouse.
OnePlus showcases an impressive gaming-focused smartphone that you probably won't be able to obtain.
You're not prepared for this gaming powerhouse.
 The Batman Part II: Release date, cast, storyline, and all the information we have at this point.
What’s the most recent update on The Batman Part II? Here’s all the information we have on the sequel to The Batman, including the release date, storyline, and cast.
The Batman Part II: Release date, cast, storyline, and all the information we have at this point.
What’s the most recent update on The Batman Part II? Here’s all the information we have on the sequel to The Batman, including the release date, storyline, and cast.
 Apple TV and Peacock Premium Plus have both seen a price reduction, courtesy of Amazon.
That's a great offer!
Apple TV and Peacock Premium Plus have both seen a price reduction, courtesy of Amazon.
That's a great offer!
AliveCor’s Kardia 12L has received its CE marking.
AliveCor's Kardia 12L, a compact AI ECG device, has obtained CE Mark certification for its introduction in France, Germany, Italy, Spain, and the UK.
