
Stockholm's BioLamina obtains a €20 million loan from the EIB.
The European Investment Bank is providing a €20 million loan to BioLamina, a Swedish biotechnology firm that supplies laminin-based cell culture matrices utilized by stem cell therapy developers globally. This funding will aid in the expansion of laminin technology production and the development of animal-free drug safety testing methods.
BioLamina, based in Stockholm, focuses on producing protein scaffolding necessary for growing stem cells intended for therapeutic use. The company has secured a €20 million loan from the European Investment Bank. According to the EIB's Stockholm office, this financial support will facilitate the further development of laminin technologies that enable advanced cell therapies and more sophisticated animal-free drug safety testing methods.
BioLamina plays a vital role in the cell therapy ecosystem by providing extracellular matrix proteins that help researchers and companies cultivate, expand, and differentiate stem cells reliably outside the human body, rather than developing therapies directly. Its main product line, Biolaminin®, consists of full-length human recombinant laminin proteins, which mimic the proteins found in natural tissue environments.
Laminins are essential structural components of the basement membrane, which lies beneath epithelial and endothelial cells, and they induce specific responses in cells, including adhesion, survival, differentiation, and organization. Most commercial alternatives use truncated or non-human versions of these proteins; BioLamina asserts that using full-length human laminins leads to more biologically relevant conditions, resulting in cell products that are more consistent, reproducible, and clinically compliant.
The potential therapeutic uses for cells cultivated on laminin matrices are extensive. These cell therapies may be effective in treating conditions such as type 1 diabetes (by generating insulin-producing beta cells), Parkinson’s disease, heart failure, acute liver failure, and cancer, among others that are currently challenging to treat or for which there is no cure.
BioLamina has partnered with Novo Nordisk on stem cell treatments for diabetes, and it collaborates with Cell X Technologies on iPSC-based therapeutic manufacturing workflows. Established in 2008 based on research conducted by Dr. Karl Tryggvason at the Karolinska Institutet and Duke-NUS Medical School, BioLamina is headquartered in Sundbyberg, part of Greater Stockholm, and employs around 100 to 117 individuals, according to various estimates.
Its main shareholders include the Swedish investment firm Bure Equity AB (through Bure Growth), Lauxera Capital Partners, the Tryggvason family, and Northislet. The company secured €19 million in equity financing in July 2024, led by Lauxera Capital Partners.
The €20 million loan from the EIB marks the company’s first publicly disclosed debt financing and aligns with the EIB's broader BioTechEU initiative, announced in December 2025, which aims to mobilize €10 billion in biotech investments during 2026-27. CEO Klaus Langhoff-Roos has articulated BioLamina’s mission as demonstrating that “full-length equals full-function,” a principle that extends to its drug safety testing products, which offer animal-free alternatives to the traditionally used animal-derived extracellular matrices in toxicology screening. This EIB loan will support both facets of the business.
Other articles
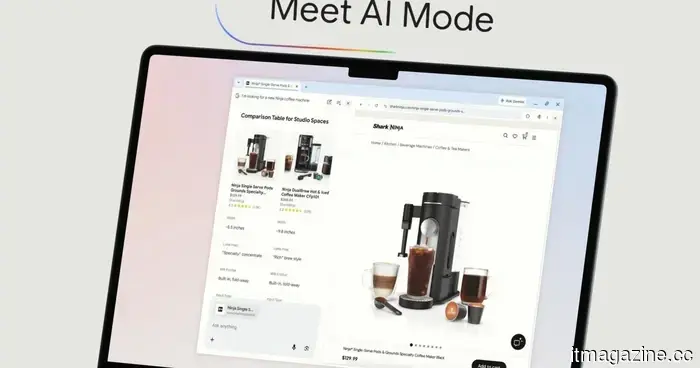 The AI mode in Chrome receives a significant enhancement aimed at reducing the need for switching between tabs.
Google's AI Mode enhancement for Chrome enables you to browse websites and conduct searches simultaneously, allowing you to pose follow-up questions without losing your spot or opening an additional tab.
The AI mode in Chrome receives a significant enhancement aimed at reducing the need for switching between tabs.
Google's AI Mode enhancement for Chrome enables you to browse websites and conduct searches simultaneously, allowing you to pose follow-up questions without losing your spot or opening an additional tab.
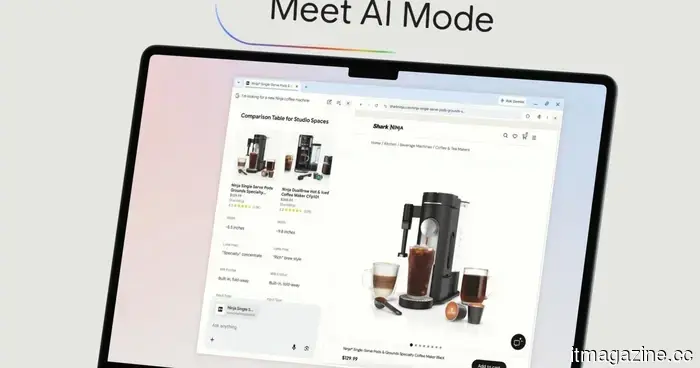 The AI feature in Chrome receives a significant enhancement to reduce the need for switching between tabs.
The AI Mode upgrade for Google Chrome allows you to browse websites and conduct searches simultaneously, enabling you to ask follow-up questions without having to lose your spot or open an additional tab.
The AI feature in Chrome receives a significant enhancement to reduce the need for switching between tabs.
The AI Mode upgrade for Google Chrome allows you to browse websites and conduct searches simultaneously, enabling you to ask follow-up questions without having to lose your spot or open an additional tab.
 DeepL introduces real-time voice-to-voice translation in over 40 languages.
DeepL has introduced Voice-to-Voice, a real-time spoken translation tool designed for meetings, discussions, and enterprise API.
DeepL introduces real-time voice-to-voice translation in over 40 languages.
DeepL has introduced Voice-to-Voice, a real-time spoken translation tool designed for meetings, discussions, and enterprise API.
 AlixLabs secures €15 million in Series A funding.
AlixLabs has completed a €15M Series A funding round supported by Navigare, Industrifonden, Global Brain, and Stephen Industries to bring its APS™ to market.
AlixLabs secures €15 million in Series A funding.
AlixLabs has completed a €15M Series A funding round supported by Navigare, Industrifonden, Global Brain, and Stephen Industries to bring its APS™ to market.
 AlixLabs secures €15 million in Series A funding.
AlixLabs has successfully completed a €15M Series A funding round, supported by Navigare, Industrifonden, Global Brain, and Stephen Industries, to bring its APS™ to market.
AlixLabs secures €15 million in Series A funding.
AlixLabs has successfully completed a €15M Series A funding round, supported by Navigare, Industrifonden, Global Brain, and Stephen Industries, to bring its APS™ to market.
 A $400 discount on the Samsung Galaxy Z Fold7 makes the most ambitious Android phone of 2025 significantly more accessible.
The Samsung Galaxy Z Fold7 is currently priced at $1,719.99 due to a limited-time offer, which provides a $400 discount from its original price of $2,119.99, and this deal is for the 512GB version that is worth waiting for. Foldable smartphones have significantly advanced in the past two generations, and the Z Fold7 strongly supports the case for the viability of this design.
A $400 discount on the Samsung Galaxy Z Fold7 makes the most ambitious Android phone of 2025 significantly more accessible.
The Samsung Galaxy Z Fold7 is currently priced at $1,719.99 due to a limited-time offer, which provides a $400 discount from its original price of $2,119.99, and this deal is for the 512GB version that is worth waiting for. Foldable smartphones have significantly advanced in the past two generations, and the Z Fold7 strongly supports the case for the viability of this design.
Stockholm's BioLamina obtains a €20 million loan from the EIB.
BioLamina has obtained a €20 million loan from the EIB to enhance its laminin protein technologies utilized in stem cell therapies and animal-free drug safety evaluations.
